- Bone Health
- Immunology
- Hematology
- Respiratory
- Dermatology
- Diabetes
- Gastroenterology
- Neurology
- Oncology
- Ophthalmology
- Rare Disease
- Rheumatology
Sandoz to Start Phase 3 Trial of Aflibercept Biosimilar
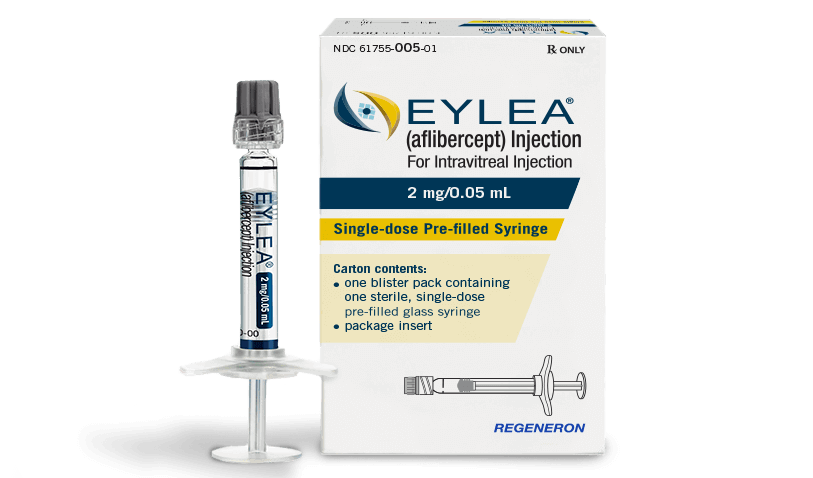
There are multiple contenders now in the race to bring the first aflibercept (Eylea) biosimilar to market in the United States and European Union.
Sandoz said it will initiate enrollment of patients in a confirmatory phase 3 efficacy and safety study (MYLIGHT) for an aflibercept biosimilar candidate. Aflibercept is used for improving visual acuity in patients with neovascular age-related (wet) macular degeneration (nAMD), diabetic macular oedema, macular oedema secondary to retinal vein occlusion, and other neovascular retinal diseases.
There are no aflibercept biosimilars on the US or EU markets. Multiple competitors are working to be among the first to tap into the market dominated by Bayer (Leverkusen, Germany) and Regeneron Pharmaceuticals (Tarrytown, New York) for the originator product (Eylea), which had US net product sales of $4.95 billion in 2020 vs $4.64 billion the previous year.
Those include Alteogen, a Republic of Korea company, that in March 2021 said it had successfully completed a phase 1 trial of an aflibercept biosimilar. In August 2020, Formycon and Bioeq, both Germany-based, initiated a phase 3 equivalency study for aflibercept candidate FYB203.
Florian Bieber, global head of Biopharmaceuticals Development at Sandoz, noted that “nAMD accounts for 10% of all age-related macular degeneration cases, but is responsible for 90% of AMD-related blindness.” Aflibercept inhibits ocular vascular endothelial growth factor–A, which promotes the growth of abnormal blood vessels in the choroid or pigmented layer of the eyeball between the outer layer of the eyeball (sclera) and the retina.
MYLIGHT investigators hope to confirm that the biosimilar candidate has equivalent efficacy and comparable safety to Eylea.
Study Details
The study is a randomized, double-blind, parallel 2-arm trial with a projected enrollment of 460 patients in 20 countries. The biosimilar candidate will be tested in patients with wet age-related macular degeneration because that is one of the most common disease types for patients who use aflibercept and because the findings can be extrapolated to other indications.
Patients with nAMD will be randomized to receive either the biosimilar candidate or the reference product for 48 weeks. The mean change in best corrected visual acuity score from baseline to week 8 will serve as the primary end point.
Sandoz is heavily invested in biosimilars. The company has 3 supportive care oncology biosimilars and 1 blood cancer biosimilar that doubles as an immunology drug. The company has 3 other immunology biosimilars and an endocrinology drug, for a total of 8 biosimilars on the market. Sandoz also has a denosumab biosimilar in phase 3 development, along with various other development-stage biosimilar candidates.
Newsletter
Where clinical, regulatory, and economic perspectives converge—sign up for Center for Biosimilars® emails to get expert insights on emerging treatment paradigms, biosimilar policy, and real-world outcomes that shape patient care.